1. Introduction and Methods to Measure Rate
Methods of Measuring Rate of Reactions
In this video, we will explore two methods to measure rate of reactions:
(1) volume of gas per unit time and,
(2) mass loss per unit time


2. Collision Theory and Factors Affecting Rate of Reactions
Collision Theory and Factors Affecting Rate
In this video, we will explore how Collision Theory is used to explain the effect of the following factors on the rate of reactions:
(1) temperature
(2) concentration
(3) pressure
(4) particle size
(5) catalysts (G3 Chemistry only)
An Analogy
For a reaction to take place, the reactants must not only collide, they must collide with the correct orientation and enough energy. This minimum energy that reactions must posses imposes a barrier to the reaction, known as the activation barrier or activation energy.
A collision that meets the above criteria is termed an effective collision. To increase the rate of a reaction,
-
We can lower the activation energy (by using a catalyst)
-
We can increase the frequency of collisions (make particles closer)
Molecular Simulation

Visit the following website to access the simulation.

Click on the “Rate Experiments” Tab
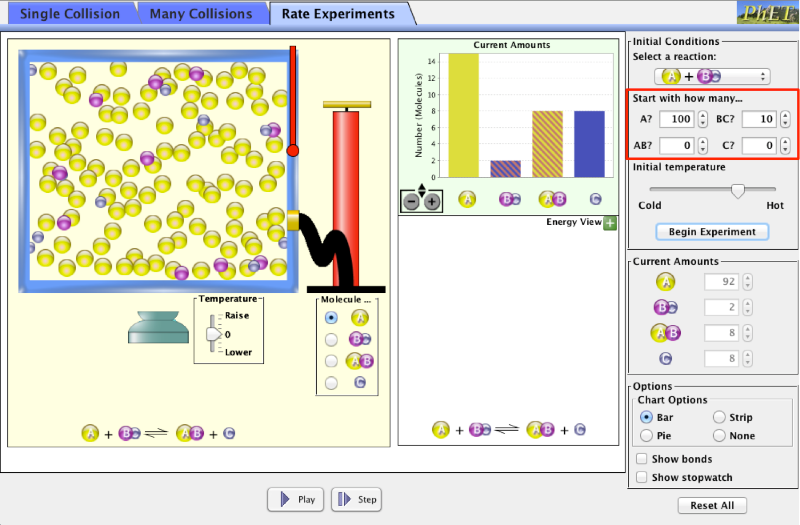
Now increase the amount of reactant A from 10 to 100. Begin experiment. Explain, in terms of the collision theory, why the rate of reaction increases when the concentration of reactant A increases.

Visit the following website to access the simulation.
Effect of Temperature on Speed of Reaction
This video shows the reaction of sodium thiosulfate with hydrochloric acid at two different temperatures. We can see that the speed of reaction increases as the temperature increases.
This can be explained using the Collision Theory. When temperature increases, reacting particles possess higher kinetic energy, they move faster and the force of collision is greater. Frequency of effective collisions between the reacting particles increases. Since frequency of effective collision is proportional to speed of reaction, speed of reaction increases
Lightstick Kinetics – Effect of Temperature
Change how brightly a lightstick glows by placing it in different temperature water baths.
Effect of Concentration on Speed of Reaction
This video shows the reaction of magnesium with hydrochloric acid of different concentrations. We can see that the speed of reaction increases as the concentration of the reactants increases.
This can be explained using the Collision Theory. When concentration increases, number of particles per unit volume increases. Frequency of effective collisions between the reacting particles increases. Since frequency of effective collision is proportional to speed of reaction, speed of reaction increases.
Lycopodium Fireball – Effect of Surface Area

After watching the video, you now have an idea on the relationship between surface area and rate of reactions. In your groups, think of how this relationship can be useful/ harmful in real life? Search for news/examples online to illustrate your point. One example you can look up is the water park explosion in Taiwan (2015).
Using the context of your example, craft an explanation for how the increase/decrease in surface area resulted in the desired/undesired change in the rate of reaction. Share your responses as a group here:
Effect of Particle Size on Speed of Reaction
This video shows the reaction of calcium carbonate in different forms with hydrochloric acid. We can see that the speed of reaction increases as the particle size of reactants decreases.
This can be explained using the Collision Theory. When particle size decreases, the total surface area exposed to the reacting particles is increased. This leads to a larger total surface area to volume ratio. The frequency of effective collisions between the reacting particles increases. Since frequency of effective collision is proportional to speed of reaction, speed of reaction increases.
Elephant's Toothpaste – Effect of Catalyst
Steve returns to The Ellen DeGeneres Show with some explosive experiments. You won't believe your eyes when Steve makes a boat float on air, creates a giant wall of foam, and sets off a massive explosion with a bouquet of balloons!
Application of Enzymes – Pectinase
Pectinase enzymes are used for extracting juice from purée. This is done when the enzyme pectinase catalyse the breakdown the substrate pectin and the juice is extracted. The enzyme pectinase lowers the activation energy needed for the juice to be produced and catalyzes the reaction.
Effect of different catalysts on rate of decomposition
Several measuring cylinders are set up each containing a little washing up liquid and a small amount of a catalyst for the decomposition of hydrogen peroxide. Hydrogen peroxide is poured into the cylinders and a foam rises up the cylinders at a rate that depends on the effectiveness of the catalyst.
3. Clock Reactions and Data Processing
Clock Reactions and Data Processing
In this video, we will explore how to measure rates of reaction using the clock reaction method and consolidate our learning by looking at a data-based question.
4. Graph Sketching
