
1. Introduction to Organic Chemistry


A Brief Guide to Types of Isomerism
In organic chemistry, isomers are molecules with the same molecular formula (i.e. the same number of atoms of each element), but different structural or spatial arrangements of the atoms within the molecule. The reason there are such a colossal number of organic compounds – more than 10 million – is in part down to isomerism. This graphic looks at the 5 main types of isomerism in organic molecules, with a more detailed explanation of each given below, as well as the reason why isomerism is important in our day-to-day lives.
*Note that stereoisomerism is not in O Level Syllabus
Functional Groups in Organic Compounds
A functional group in chemistry is the part of the molecule that gives it its particular reactivity; simple molecules that contain the same functional group in their structure can be expected to react in similar ways. More complicated chemical molecules may contain more than one functional group within their structure, which can sometimes affect the chemical reactions they undergo. The names of organic molecules are systematic references to the functional groups within the molecule, and can thus be used to identify these.
Although we only learn a few functional groups at O Levels, there are many more functional groups out there, which makes Chemistry so diverse an interesting! This infographic lists 21 common functional groups in nature.
Tutorial – Homologous Series and Nomenclature
In this video, we describe a homologous series as a group of compounds with a general formula, similar chemical properties and showing a gradation in physical properties as a result of increase in the size and mass of the molecules, e.g. melting and boiling points; viscosity; flammability. We will also learn how to name and draw various organic molecules.
2. Fuels and Crude Oil
Tutorial – Fuels and Crude Oil
At the end of this video, you should be able to
a) name natural gas, mainly methane, and crude oil as non-renewable sources of energy
b) describe crude oil as a mixture of hydrocarbons and its separation into useful fractions by fractional distillation to yield fractions which have competing uses as fuels and as a source of chemicals
Tutorial – Biofuels
At the end of this video, you should be able to
a) describe biofuel (exemplified by bioethanol from sugarcane) as a renewable alternative to natural gas and crude oil
b) describe how biofuel, when compared to fossil fuels, can be more environmentally sustainable in terms of carbon dioxide emission
Can 100% renewable energy power the world?
Every year, the world uses 35 billion barrels of oil. This massive scale of fossil fuel dependence pollutes the earth, and it won’t last forever. On the other hand, we have abundant sun, water and wind, which are all renewable energy sources. So why don’t we exchange our fossil fuel dependence for an existence based only on renewables? Federico Rosei and Renzo Rosei describe the challenges. Lesson by Federico Rosei and Renzo Rosei, directed by Giulia Martinelli.
Biofuels in Singapore
Singapore has started to adopt renewable fuels in the aviation and shipping industries. For example, Singapore Airlines has piloted the use of blended sustainable aviation fuel for its flights.
The Maritime Port Authority of Singapore has also been facilitating the use of biofuels. Over 140,000 tonnes of biofuels blends have been supplied across more than 90 biofuels bunkering operations. Watch the video to learn how Singapore develops the national standard for marine biofuels.
Sustainable Aviation Fuel
Sustainable aviation fuel (SAF) is a critical enabler for the airline industry to reach new heights in achieving carbon net-zero emissions by 2050. Yet, travellers do not know much about SAF, how it is produced, and why there is not enough of it.
Economic, Environmental & Social Effect of Biofuels
Learn the basics about the economic, environmental and social effects of biofuels as part of the fuels chapter within environmental chemistry.
Fracking explained: opportunity or danger
There has been a lot of talk about a controversial method of extracting natural gas: Hydraulic fracturing or fracking. Put simply, fracking describes the recovery of natural gas from deep layers inside the earth. In this method, porous rock is fractured by the use of water, sand and chemicals in order to release the enclosed natural gas.

Oil Spill Clean-Ups Using Fruits & Oils
An oil spill is the release of a liquid petroleum hydrocarbon into the environment, especially marine areas, due to human activity, and is a form of pollution.
Julian Silverman is currently researching how compounds obtained from fruits and oils can be utilised in a number of applications, including helping to clean up oil spills at sea. Here, he explains the chemistry behind the process and how it works.

The Chemistry of Petrol & The Tetraethyl Lead Story
Petrol contains hydrocarbons with chains between five to twelve carbon atoms long, with diesel’s chains being slightly longer at ten to fifteen atoms. Diesel also contains more energy than petrol per litre, making it a more efficient fuel, albeit a more expensive one.
Petrol and diesel engines also work in slightly different ways. In petrol engines, knocking can be a problem. to prevent engine knocking, scientists have added a range of compounds to petrol over the years. You’ve probably come across the octane rating of fuels before – this is essentially a measure of how well the fuel avoids the knocking problem.
3. Alkanes

Credits: David Klein
Isomerism in alkanes
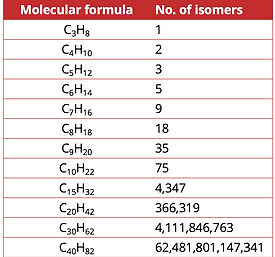
Alkanes with more than three carbon atoms can be arranged in numerous different ways, forming different structural isomers (branched alkanes).
For an alkane, the number of possible isomers increases with increasing molecular size..
When drawing the constitutional isomers of an alkane, make sure to avoid drawing the same isomer twice.
Can you draw the five isomers of hexane?
Tutorial - Isomers
At the end of this video you should be able to
• define the term isomer
• draw and name isomers
Alkanes are generally unreactive but undergo combustion
Alkanes are generally unreactive due to the strong C–C and single C–H bonds. Given the lack of chemical reactivity, there are few applications for alkanes other than them serving as a good fuel in combustion. Much research has been devoted to the design and synthesis of new reagents and catalysts that can activate the C–H bond (i.e. to make it more reactive). C–H activation chemistry could enable the conversion of cheap and abundant alkanes into valuable functionalized organic compounds.
Alkanes are highly flammable. Part of a Queensland river bubbling with methane gas has burst into flames after being ignited by a Greens MP, who blames nearby coal seam gas (CSG) operations for the tragedy in the Murray-Darling Basin.
Ongoing research has identified several scenarios that could be contributing to the seeps including the natural geology and faults (formed tens-of-millions of years ago), natural events such as drought and flood cycles as well as some human activity, which includes water bores and coal seam gas operations.

Alkanes undergo substitution to form alkyl halides
This is a very important reaction as it converts underactive alkanes into alkyl halides that are reactive compounds that undergo a wide range of reactions. Alkanes react with molecular chlorine and bromine on irradiation with UV light to form chloroalkanes and bromoalkanes, respectively.
Think about this:
-
Using bond energies, can you explain why it is not practical to use fluorine and iodine in substitution reactions involving alkanes?
-
Why are alkyl halides (such as Halon 1211 and Halon 1301) more effective than carbon dioxide in extinguishing fires? Are there any disadvantages of using alkyl halides?
3. Alkenes

Ethene and the Flavr Savr Tomato
Ethene is a plant growth hormone that promotes the ripening of fruits such as apples, bananas, and tomatoes. Ethene is produced in plants from S-adenosylmethionine (SAM).
Recently, several varieties of tomatoes have been genetically engineered to ripen more slowly. One way of doing this is to alter the gene sequence of the tomato so that production of the enzyme ACC synthase is decreased, which, in turn, decreases the production of ethene. This makes the fruit last longer in shipment and, on arrival, the genetically engineered fruit is treated with ethene to induce ripening.



What makes certain molecules coloured?
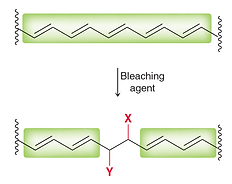
Transition metals form many ionic compounds that are coloured, such as copper(II) chloride or iron(III) chloride. But covalent compounds can be coloured if they contain a special arrangement of C=C double bonds.
Lycopene is responsible for the red color of tomatoes while β-carotene is responsible for the orange color of carrots. Both compounds are colored because they possess many alternating C–C single bonds and C=C double bonds, a property known as conjugation.
The Power of Bleach
Water and bleach: historically, this pair go together pretty well, namely in the world of cleanliness. Bleach is a remarkably caustic base that, when diluted with water, can clean stains from whites and linoleum. However, apart from their similarly clear appearance, water and bleach are very dissimilar. Bleach is very alkaline, but water is a neutral pH. Chemically, that is saying a lot. But what does that have to do with vivid colors?
Common household bleach, such as Clorox, work by oxidizing the double bonds. This disrupts the alternating pattern of C–C and C=C bonds. When a stain is bleached, it has not been washed away. Rather, it has just been chemically altered so that it can no longer be seen.

Test for unsaturation
Add aqueous bromine in the absence of UV light. If an alkene is present, the reddish-brown aqueous bromine decolourises rapidly.
Making Margarine in the Lab
NileRed shows us how to use a laboratory procedure to make margarine from olive oil. The palladium on carbon (Pd/C) catalyst used here works in the same way as the nickel catalyst we learn in our syllabus.
Many Chemistry techniques and concepts are covered in this synthesis video, such as reaction of acids to generate hydrogen, separation and purification techniques.
The Truth About Fats and Nutrition
First low fat was in, now high fat is in—what kind of diet is actually the best for you? Join Hank Green and learn what we DO know about fat chemistry in this new health-focused episode of SciShow!
Why are PHOs banned in Singapore?
Singapore has banned partially hydrogenated oils (PHOs), a major source of artificial trans fat found in snacks, baked goods and fast food. Channel NewsAsia's Jalelah Abu Baker explains what partially hydrogenated oils are, and how the ban affects you.
Cracking a Hydrocarbon
Liquid paraffin (a mixture of alkanes of chain length C20 and greater) is vaporised and passed over a hot pumice stone catalyst. A gaseous product is obtained which is flammable and which will decolorise bromine water and acidified manganate(VII) ions. The same apparatus and method can be used to dehydrate ethanol.

Chemistry of rancidity & Olive oil
When olive oil is kept for long or not stored properly, it becomes rancid, that is, having an unpleasant smell or taste.
This is caused by the hydrolysis and/or oxidation of the fats into short-chain aldehydes and ketones which are objectionable in taste and odor.
To prolong the lifespan of olive oil, it should be stored in a cool and dark place, and preferably in a dark bottle.
Addition of hydrogen and making margarine
Learn how to harden vegetable oils through a chemical process demonstrated on the example of margarine.
Which cooking oil contains the most polyunsaturated fats?
How can aqueous bromine be used to find out which cooking oil contains the most unsaturated fats?
What type of reaction takes place? What happens to the reddish-brown bromine?
Why is the product more viscous than the reactant?

A Guide to the Different Types of Fat
Fat is an important nutrient in our diets, but there’s a lot of talk of different types of fats, and whether these types are beneficial or harmful to our health. These different fat classifications have their roots in chemistry – and chemistry can also help explain their effects. This graphic takes a look at the different classifications, their sources, and briefly about how they act in our body.
4. Alcohols
What are Alcohols? (Enrichment)
Note: Hydrogen bonding, primary, secondary and tertiary alcohols are not in syllabus.
Bouncing Bubbles
There's something magical about a bubble. It's just a little puff of air trapped in a thin film of soap and water, but its precise spherical shape and beautiful swirling colors make it a true wonder of science. A bubble's life expectancy is usually measured in seconds unless you know how to make a SUPER BUBBLE! The magic ingredient lies in a molecule with three hydroxyl functional groups!
The Whoosh Bottle
A mixture of alcohol and air in a large polycarbonate bottle is ignited. The resulting rapid combustion reaction, often accompanied by a dramatic ‘whoosh’ sound and flames, demonstrates the large amount of chemical energy released in the combustion of alcohols.
Burning Ethanol
Before watching the video, make predictions to the following questions:
-
What changes in mass would you observe?
-
What changes in the Bromothymol blue would you observe?
-
Where are atoms moving?
-
Where does the energy come from?
Good link to conservation of mass, particle theory, acids and bases, and energy changes.
Test for Alcohols
Add acidified potassium manganate(VII) to the unknown and heat. If the unknown is an alcohol, the purple potassium manganate(VII) turns colourless.
Note: In JC or more advanced Chemistry courses, you will learn that not all alcohols react with acidified potassium manganate(VII), but at O Levels, we just take it that all alcohols will give a positive test.
Making Alcohols by Fermentation and from Ethene
Learn the basics about making alcohols by fermentation and from ethene, as part of organic chemistry.
5. Carboxylic Acids and Esters
Carboxylic Acids, Typical Acids and Esters
Learn the basics about Carboxylic and Typical acids, and also what are esters and what are they used for? How do all these terms relate to one and other? Find out more in this video!
How to make Grape Flavour | Methyl anthranilate
NileRed shows us how to use a laboratory procedure to make the ester methyl anthranilate. Concentrated sulfuric acid serves as a catalyst for the reaction.
We learnt that esters are an oily liquid and here you see it being extracted using an organic solvent, dichloromethane (DCM)
Many Chemistry techniques and concepts are covered in this synthesis video.
Table of Esters and their Smells
Created by James Kennedy. More infographics at: https://jameskennedymonash.wordpress.com/

6. Polymers
Natural Polymers
Learn the basics about natural polymers. What are natural polymers? where are they found and how are they structured?
Teflon was invented by accident
The man who accidentally invented Teflon was Dr. Roy Plunkett. After receiving his BA, MS, and eventually PhD in organic chemistry, Dr. Plunkett took a job with DuPont, in Jackson New Jersey. He was subsequently assigned to work on synthesizing various new forms of refrigerant, trying to find a non-toxic alternative to refrigerants like sulfur dioxide and ammonia.
Ultimate Gooey Slime
A solution of polyvinyl alcohol (PVA) can be made into a slime by adding borax solution, which creates crosslinks between polymer chains.
Tutorial - Addition Polymersiation
This video teaches you how to draw the structural formula (or a number of repeating units) of a polymer from a given monomer and vice versa.
Tutorial - Condensation Polymersiation
This video teaches you how to draw the structural formula (or a number of repeating units) of a polymer from a given monomer and vice versa.
Nylon Synthesis
Nylon is a condensation polymer formed by reacting two monomers, one is a diamine and the other is a dicarboxylic acid. The result is a polymer containing the amide linkage.
Polymer Chemistry in Bowling Balls
In materials science or polymer chemistry, a resin is a soft solid or highly viscous substance, usually containing prepolymers with reactive groups. Such viscous substances can be plant-derived or synthetic in origin. They are often mixtures of organic compounds.
Many bowling balls contain reactive resin shells (used to help improve ball friction) comprising of a mixture of urethane and a proprietary ingredient that generates surface porosity while the polymer base cures. Ball makers mix isocyanates and polyols to form the base urethane resin.

Acetone Dissolves Styrofoam (Polystyrene)
Have you ever seen a foam cup appear to "melt away" in acetone? Foam cups, bowls and containers are made of a lightweight but strong material – expanded polystyrene. How then does acetone transform this substance to a blob of goo?
Making Soap
NileRed shows us how to use a laboratory procedure to make soap.
Many Chemistry techniques and concepts are covered in this synthesis video.
Weaving the Future With Fishing Net Recycling
To help reduce the "ghost" marine debris that does not decompose, those involved in Japanese fisheries have launched a project to collect and recycle used fishing nets.
Watch the documentary from NHK World and think of the following questions:
• What are the environmental concerns of plastics depicted in the video?
• Should we ban plastics?
• What makes recycling of plastics a challenge?
Turning Plastic Bottles into Clothing
When plastic is recycled, it is melted, cooled, pulled into long, thin strands and cut into pellets to be made into new products. The chemical composition of the plastic remains unchanged.
What happens to plastic bottles when you recycle them? Do they get made into fresh bottles, or do they become something else?


