
1. Introduction
Misconceptions about Temperature
In this video interview, Veritasium uncovers the public's ideas about temperature, heat transfer, and properties of materials.
Misconceptions about heat
When we touch something that is hot or cold, what are we actually sensing? Is it the temperature of the object, or the rate at which heat flows between the object and our hand?
The Science of Firewalking
For a long time, people have been amazed at the ability of Eastern mystics to walk across beds of glowing coals without any apparent discomfort. Even in Singapore, about 4,500 Hindu devotees gather each year at the Sri Mariamman Temple along South Bridge Road to take part in Thimithi, the annual fire-walking festival.
How can this be possible?
2. System and Surroundings
-
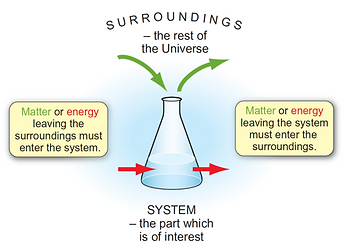
The system refers to the specific part of the universe that is of interest to us, i.e. the sample of matter or the reaction that is being studied.
-
The surroundings are the rest of the universe outside the system.
-
This definition allows you to keep track of where energy and matter move during a reaction.

3. Enthalpy (Enrichment)
What is Enthalpy?
In order to fully understand why we talk about enthalpy change and not absolute enthalpy, we will need to dive deeper into physical chemistry beyond the O Level syllabus.
What triggers a chemical reaction?
Chemicals are in everything we see, and the reactions between them can look like anything from rust on a spoon to an explosion on your stovetop. But why do these reactions happen in the first place? Kareem Jarrah answers this question by examining the two underlying forces that drive both endothermic and exothermic chemical reactions: enthalpy and entropy.
Calorimetry, food and metabolism
The energy content of foods can be determined from experiments using a bomb calorimeter. While the bomb calorimeter gives a good measure of the energy content of foods, it does not, of course, take into account how food is processed in the body. Different people have different rates of consuming energy and turning food into energy—their metabolisms differ.
4. Exothermic and Endothermic Reactions
Japan's self-warming bento box
Just pull a string and the box heats up like an oven. You’ll even see hot steam coming out! (Note that the guy in the video incorrectly referred to it as "smoke")
Here’s how it works: Underneath the food is a container of tiny white pieces of calcium oxide. Pulling the string releases water which mixes with the coal, causing a the chemical reaction.
Homemade Hand Warmer
If you’ve ever spent time outdoors in the cold, there’s a good chance you’ve used or seen chemical hand warmers. If you want to experience the science inside of those disposable plastic bags, then try your chilly hand at the Homemade Hand Warmer experiment. You can witness how an everyday chemical reaction like rusting can be used to keep your digits from falling off.
Hot ice: Stalagmites from supersaturated solutions
Hand warmers containing a liquid in a flexible plastic shell with a small, curved metal disc are commonplace. The warmer is activated by inverting the disc, which presents nuclei to support the formation of crystals from a supersaturated solution. Needles propagate out from the nucleation site and within a matter of seconds the contents have solidified. The process is exothermic, generating heat.
5. Bond Breaking and Bond Making
No content added









